Blog: Business
Manufacturing LIMS Webinar Draws a Crowd
24th January 2022

One of the best attended Autoscribe Informatics’ educational webinars in 2021 was the webinar on LIMS for manufacturing environments, titled: ‘Unraveling the role of LIMS in a Manufacturing Environment’.
A recording of this webinar can be found here:
https://register.gotowebinar.com/register/3221163763392925454?source=website
Having to collect and manage test data in manufacturing environments is key to ensuring product quality. This is certainly the case for Autoscribe customers working in diverse industries such as Pharmaceuticals, Medical Devices, Food & Beverage, Electric Vehicles and Consumer goods. Examples of how Autoscribe’s Matrix LIMS helps them manage product quality can be found in the case studies section on our website.
The recent webinar highlighted the need for efficient information management systems to support an effective manufacturing environment, highlighting examples of the cost to industry and human life when things go wrong. One example given was a 2013 McKinsey report which estimated that non routine quality events would cost the medical device industry $2.5 to $5.0 billion, with single recalls costing up to $600 million. Another example identified contaminated food as a possible cause of up to 420,000 deaths worldwide per annum.
![Making COVID 19 Vaccine]()
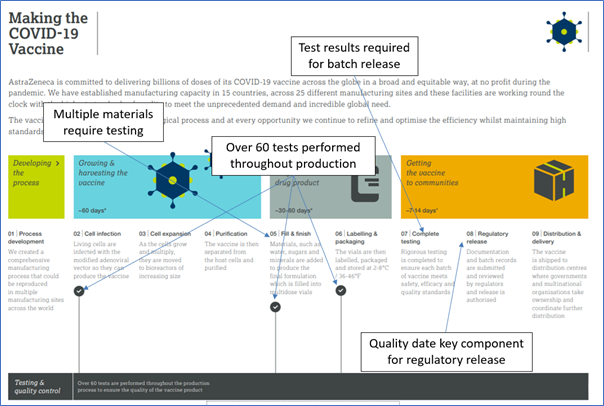
Example of Covid-19 Manufacturing Flow used in the webinar to highlight production steps [Source Astra-Zeneca{1}]
Using the manufacture of a Covid-19 Vaccine as an example, the role of LIMS in the various manufacturing stages was discussed. Testing of the raw materials as well as intermediates and the final product highlighted the key role laboratory test results have in the manufacture and release of product. LIMS was briefly compared to other IT systems such as ERP/MRP systems to illustrate how typical LIMS functionality supported both quality control and general laboratory management. Such functionality incudes, for example, instrument calibration and maintenance modules to ensure only suitably checked equipment is used, inventory management and runsheet templates that define the layout of instrument or analyst worklists including the positioning of QC samples (controls, spikes, duplicates etc.).
Topics in the webinar included:
- The impact of manufacturing quality failures
- The batch manufacturing data environment and the role of LIMS within that environment
- Drawing a line between ERP/MRP and LIMS
- The essential ingredients of a manufacturing LIMS
- Protecting your investment, the need for flexibility and longevity in your LIMS
Feedback from those attending the webinar was particularly positive and included the following comments:
- “I just want to thank everyone who attended and prepared this wonderful webinar. Great job!”, LIMS Business Analyst, Ukraine
- “Great job on the webinar. Glad I attended”, Senior Metrologist, USA
- “Aloha and Mahalo, I learned a lot and like to continue to learn more. Great Job!”, Tissue Procurement Specialist, Honolulu, USA
Like all webinars in Autoscribe Informatics’ educational series the webinar is available for download from our website where you can also find additional information about our laboratory informatics products and services.