Blog: Regulatory
FDA Inspection Checklist Emphasizes "Robust" Environmental Monitoring Systems
25th January 2017

To help US Food and Drink manufacturers prepare for US FDA inspections the Food Industry Council has just released an FDA Inspection Checklist. Since the Food Safety Modernization Act (FSMA) came into force the FDA has worked hard to conduct in-depth inspections regarding the preventive strategies that food and drink manufacturers have in place. The FDA Inspection Checklist provides a useful starting point to prepare for such an inspection and to check that you have the required controls and checks in place.
Three areas stand out as being particularly significant:
1. Finalize Written Food Safety Systems: Ensure that the company’s written food safety systems are finalized, and designated employees are familiar with the plans.
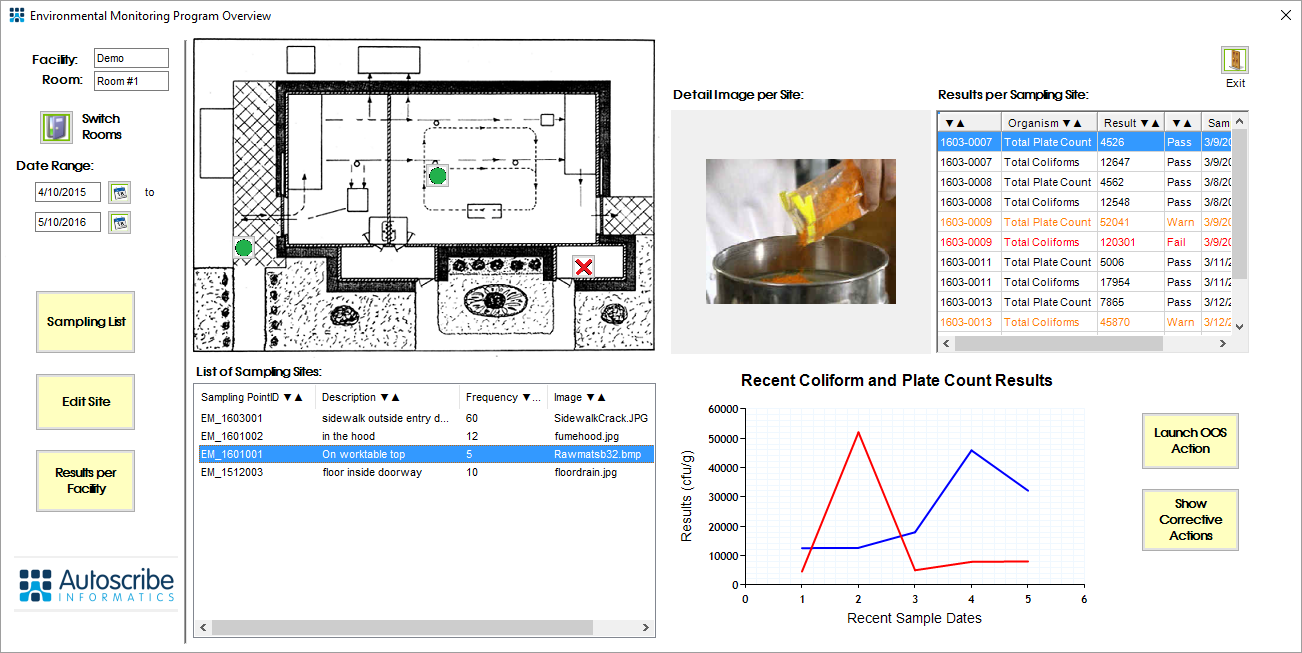
The Matrix Gemini Environmental Monitoring solution is specifically designed to help document sanitation programs and your preventive control plan. Environmental Monitoring maps sample point locations as well as sample frequency and tracks microbial test results over time. Employees who use such a system habitually drive best practices and automatically follow a documented food safety system that records data over time, enabling recall and more detailed investigation should a sample fail.
2 Ensure Ease of Records Access: Ensure that the supporting records for each of the programs are organized and maintained so data can immediately be retrieved.
The program of routine monitoring of sample points within the Matrix Gemini Environmental Monitoring solution builds data over time which can be instantly recalled should the need arise. During the setup phase, a sanitation expert/microbiologist will set up sample point locations along with the frequency of testing required. Typically a standard operating procedure is developed which will increase testing and start ‘out-of-specification’ actions if abnormal microbial contamination is detected. The Matrix Gemini Environmental Monitoring solution has this methodology built into its flow as standard. It visually shows sample point locations and links them to the test results obtained. Test data may be displayed graphically showing result trends over time, highlighting variance and peaks in data.
3 Document Corrective Actions Properly: Ensure that in the event there is a deviation, you are documenting all corrective actions correctly.
Environmental Monitoring solutions like Matrix Gemini allow investigators to quickly focus on sample point failures and the ‘out-of-specification’ actions that were initiated to investigate and resolve these failures. Typically three months of data is requested around these sample points, though up to two years’ worth of data could be requested. This data can be instantly pulled from the database as a report for further investigation.
FDA investigators will be most interested in what happens in the event of a failure and what learning gets incorporated into your regular regime. What happens when an ‘out-of-specification’ result is obtained is the crux of preventive testing regimes. Actions might include changing sanitation methods, increasing test frequency or locations in areas of concern, segregating traffic patterns, re-training staff and so forth. Some of these actions, such as increasing test frequency, can be automated. All actions must be clearly documented, which can be done by adding appropriate records directly into the Matrix Gemini Environmental Solution. This captures the actions that each quality improvement cycle needs to discover the likely root cause of any problems and how they may be avoided in the future.
All corrective actions should identify the root cause of the deviation, actions are taken to prevent recurrence and, if product safety is not affected, a written conclusion (supported by factual and scientific data) that the deviation “does not create an immediate food safety issue.”
The emphasis should always be on preventive actions to remove potential points of failure before issues get into the final delivered products causing stock loss and costly recalls.
Conclusion
The FDA Inspection Checklist is a valuable aid to all food and drinks sanitation experts. Many of the points it makes just follow best practices. The Matrix Gemini Environmental Solution provides a practical way for food and drinks companies to document their sanitation/safety programs and instantly show written evidence of both testing and corrective actions when the FDA comes knocking.